
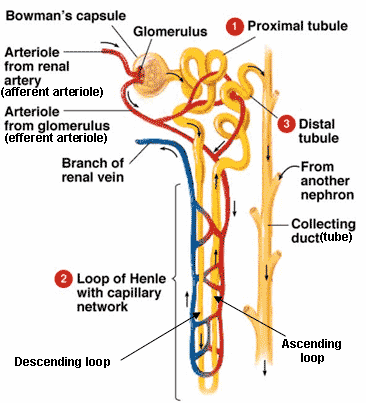
Vinaiphat A, Charngkaew K, Thongboonkerd V (2018) More complete polarization of renal tubular epithelial cells by artificial urine. In: Physiological assessment of coronary stenoses and the microcirculation. Heinonen IHA, Sorop O, Merkus D, Duncker DJ (2017) Comparative physiology and pathophysiology of the coronary circulation. įitzgerald KA, Malhotra M, Curtin CM et al (2015) Life in 3D is never flat: 3D models to optimise drug delivery. Expert Opin Drug Metab Toxicol 4:1065–1074Ĭhoucha Snouber L, Jacques S, Monge M et al (2012) Transcriptomic analysis of the effect of ifosfamide on MDCK cells cultivated in microfluidic biochips. Laverty HG, Benson C, Cartwright EJ et al (2011) How can we improve our understanding of cardiovascular safety liabilities to develop safer medicines? Br J Pharmacol 163:675–693ĭreisbach AW, Lertora JJL (2008) The effect of chronic renal failure on drug metabolism and transport. Tomlinson LA, Abel GA, Chaudhry AN et al (2013) ACE inhibitor and angiotensin receptor-II antagonist prescribing and hospital admissions with acute kidney injury: a longitudinal ecological study. Lameire NH, Bagga A, Cruz D et al (2013) Acute kidney injury: an increasing global concern. įanos V, Cataldi L (2001) Renal transport of antibiotics and nephrotoxicity: a review. Hoste EAJ, Bagshaw SM, Bellomo R et al (2015) Epidemiology of acute kidney injury in critically ill patients: the multinational AKI-EPI study. īonventre JV, Yang L (2011) Cellular pathophysiology of ischemic acute kidney injury. Mahmud HM, Mahmud SM (2017) Acute kidney injury in ureteric stones: single Centre short term analysis. Makris K, Spanou L (2016) Acute kidney injury: definition, pathophysiology and clinical phenotypes. Toxicol 10:395–408Ĭoresh J, Selvin E, Stevens LA et al (2007) Prevalence of chronic kidney disease in the United States. įisel P, Renner O, Nies AT et al (2014) Solute carrier transporter and drug-related nephrotoxicity: the impact of proximal tubule cell models for preclinical research. Įssig M, Terzi F, Burtin M, Friedlander G (2001) Mechanical strains induced by tubular flow affect the phenotype of proximal tubular cells. Raghavan V, Rbaibi Y, Pastor-Soler NM et al (2014) Shear stress-dependent regulation of apical endocytosis in renal proximal tubule cells mediated by primary cilia. Subramanya AR, Ellison DH (2014) Distal convoluted tubule. Jones G (2012) Metabolism and biomarkers of vitamin D. Meyer C, Dostou JM, Gerich JE (1999) Role of the human kidney in glucose counterregulation. Yeung CK, Himmelfarb J (2019) Kidneys on chips: emerging technology for preclinical drug development. This book chapter attempts to showcase the advancement of the kidney-on-a-chip model to mimic the kidney’s in vivo functioning and understand the cellular and molecular pathways in the kidney. The efficiency of kidney-on-a-chip models tends to envisage that successful application in the engineering of glomerulus-on-a-chip model can provide a blueprint for novel personalized organ chips and lead to ‘body-on-a-chip’ models in the future. Recent advances in kidney-on-a-chip models in the past decade could be screened and utilized for drug screening to evaluate the applicabilities of new therapies and models to understand disease mechanisms better.
It could be determined whether kidney-on-a-chip models can predict clinical outcomes before the severe stage and represent human-relevant physiology. For the advancement of kidney-on-a-chip models, there is a prime requirement to do rigorous validation. The Kidney-on-a-chip field is proving to be exponential helpful technology with the ability to revolutionize disease diagnosis, drug toxicology, and novel drug discovery.


 0 kommentar(er)
0 kommentar(er)
